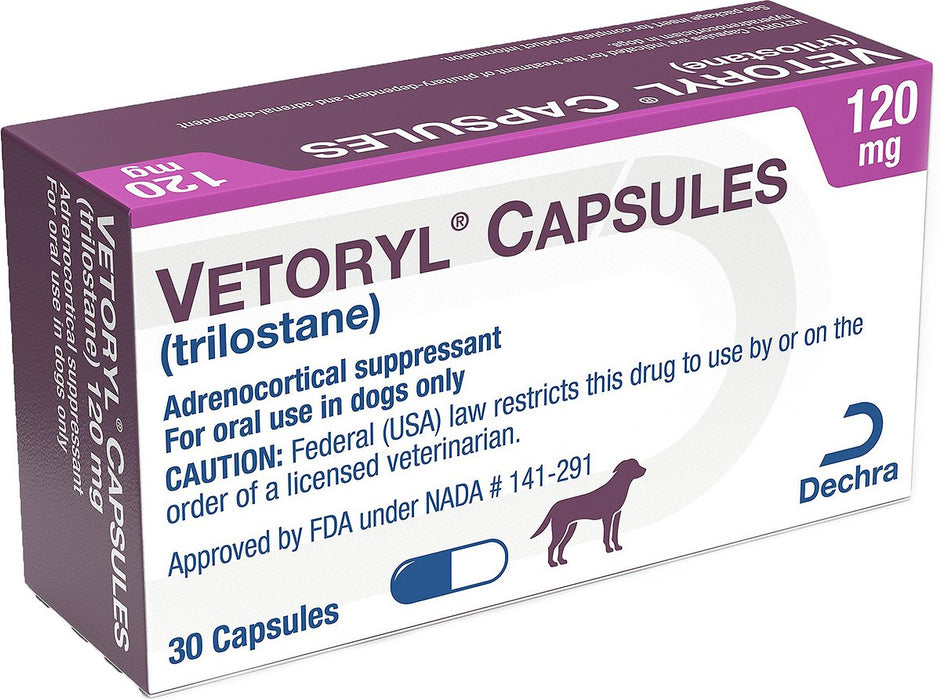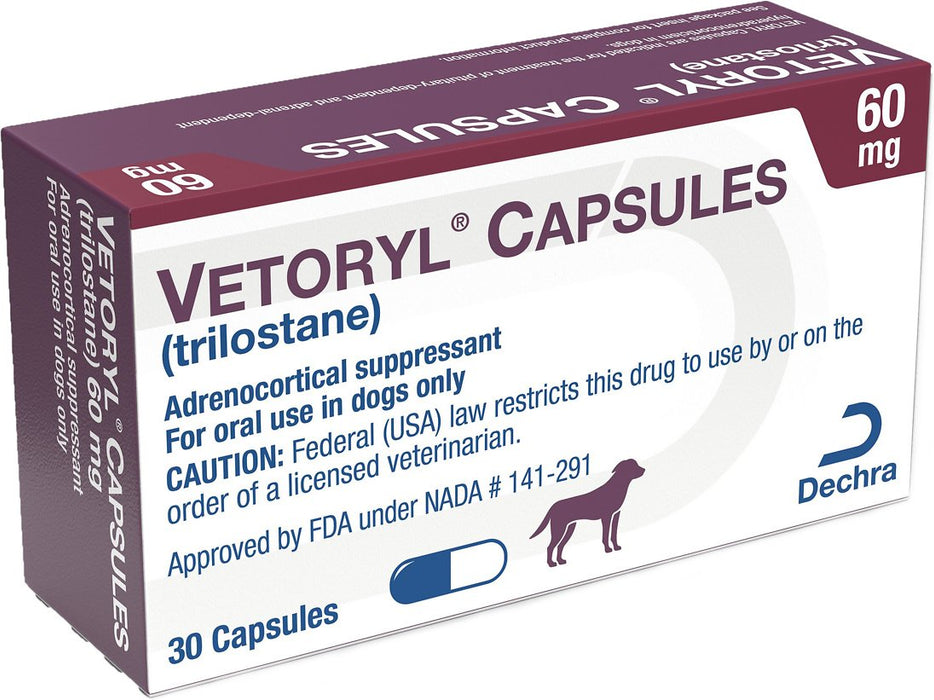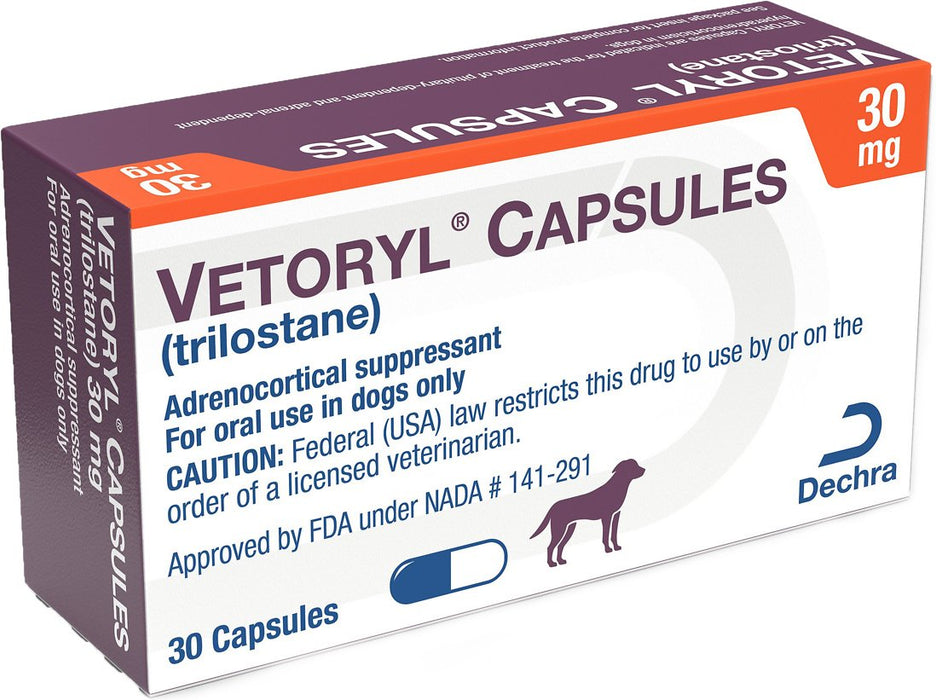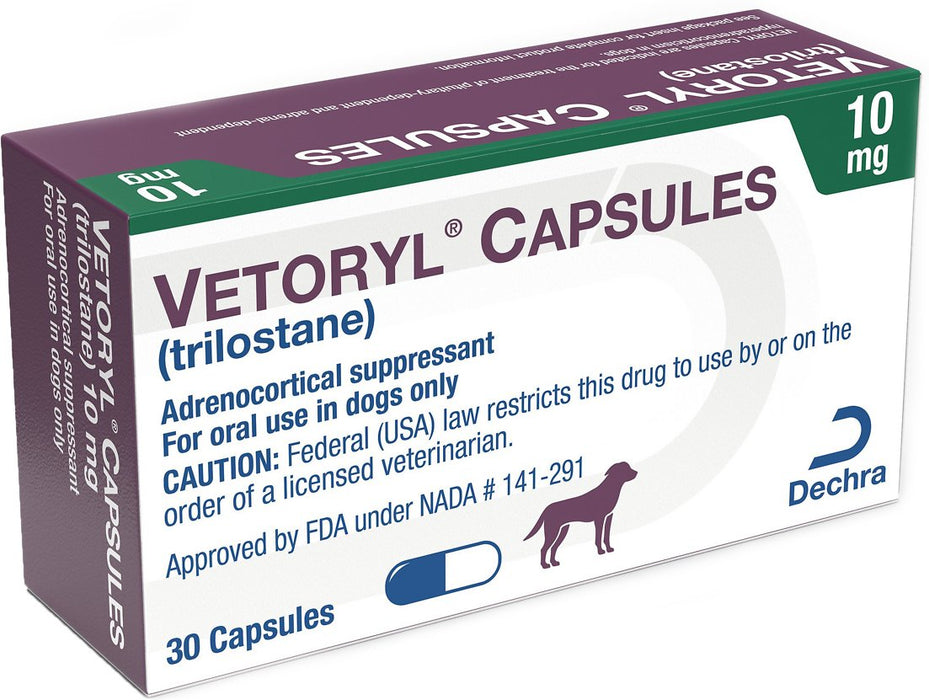
Vetoryl Capsules for Dogs
by Dechra
This item requires prescription approval.
Prescription medications are currently limited to CA residents. More states coming soon!!
Original price
$74.57
-
Original price
$249.28
Original price
$74.57
$74.57
-
$249.28
Current price
$74.57